Overview of the DAFM Animal Health Division (AHD)
Organisational structure of the DAFM Food Industry Development Division (FIDD)
|
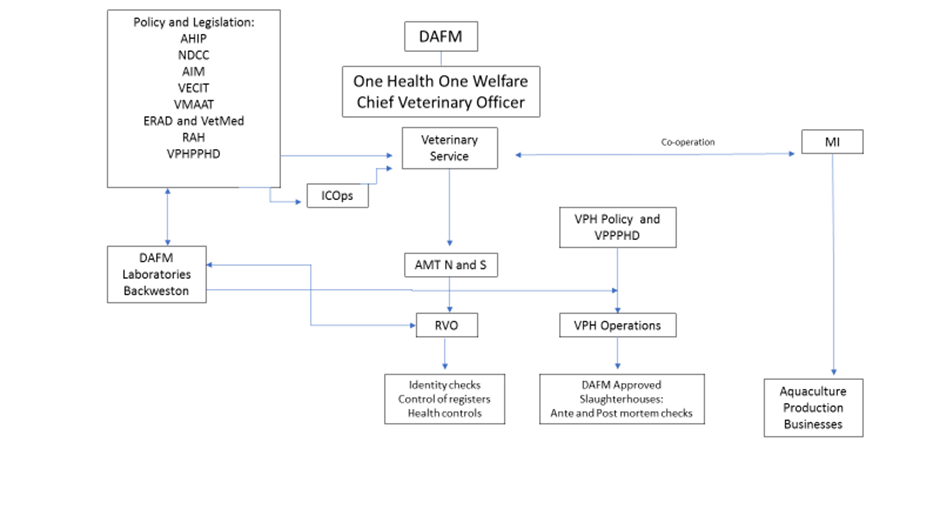
"AHIP" - Animal Health & Import Policy Division "AIM" - Animal Identification and Movement "AMT" - Area Management Team North and South "DAFM" - Department of Agriculture Food and Marine "ERAD" - Eradication "ICOps" - Import Control Operations "MI" - Marine Institute NDCC" - National Disease Control Centre |
"RAH" - Ruminant Animal Health "RVO" - Regional Veterinary Office "VECIT" - Veterinary Export; Certification, and International Trade "VMAAT" - Veterinary Medicines, AMR, ABP and TSEs "VPH" - Veterinary Public Health "VPH Ops" - Veterinary Public Health Operations "VPHPPH" - Veterinary Public Health Pig and Poultry Health |
The Veterinary Reporting Structure of DAFM, One Health, One Welfare
.png)
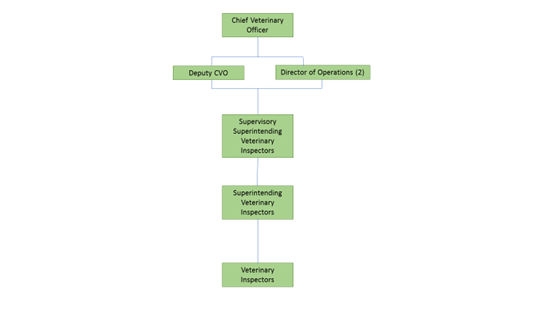
The Veterinary Service has a pyramidal management structure under the chief Veterinary Officer (CVO) and operates within the One Health, One Welfare function of the Department. The Veterinary Public Health Operations (VPHOps) is divided into seven veterinary public health regions, each under the supervision of a Regional Superintending Veterinary Inspector (SVI). The Fish Health Unit (FHU) of the Marine Institute (MI) approves aquaculture establishment (as defined in the Directive) and implements risk-based surveillance on all finfish and shellfish aquaculture sites in the country. Risk based surveillance visits are carried out by the Veterinary Service co-ordinated by Area Management Team (AMT) North. They carry out the work on behalf of the MI under a service level agreement between the MI and DAFM.
Areas of official controls as defined by Art 1(2) of Regulation (EU) 2017/625
- Article 1(2)(d) Animal health requirements
- Article 1(2)(f) Welfare requirements for animals
Delegation of official controls
There are no official controls delegated to control bodies.
Responsibility for official controls on imports is shared with the Sea Fisheries Protection Authority (SFPA) and MI.
Scope of responsibility of the AHD for the official control of food
The Veterinary Service advises the Minister of Agriculture on animal health, animal diseases, zoonoses and public health issues relating to food and products of animal origin. It also assists in the preparation, implementation and enforcement of EU and national legislation, implements control measures to protect the health of animal and human populations and provides certification for animals and animal products intended for export.
The VPHOps supervises slaughterhouses, meat processing plants, drinking milk pasteurisation establishments and egg product facilities. DAFM is responsible for the control of the importation of live animals (including live fish), products of animal origin and certain products of non-animal origin from non-EU countries at border control posts (BCPs).
The specific objectives of DAFM for controls of animal health and welfare are as follows:
- Ensure compliance with EU and national legislative requirements
- Improve the health status of farmed animals
- Ensure the welfare of animals on farm, during transport and at slaughter.
- Ensure food safety through controls in medicines, residues and zoonotic diseases
- Ensure feed and food safety through the control of TSEs and ABPs
- Apply controls regarding EU and international movement of animals
- Ensure contingency planning and control of former OIE list A diseases
- Develop contingency plans and mutual assistance arrangements
- Maintain a well-trained staff to perform official controls
- Ensure that Products of Animal Origin (POAO) and live Animals entering the European Union meet specific import conditions laid down in European Union legislation.
The DAFM Statement of Strategy and Action Plans, as well as Food Vision 2030 are available from DAFM's website.
Breakdown of Official Control Responsibilities by Division
| Division | Official Controls |
Link to DAFM Website for more information |
|
Animal Health and Import Policy (Animal Health Division) |
Registration of holdings
Pet travel Import controls |
Importing Animal and POAO |
| Animal Identification and Movement |
Bovine ID & registration (ERAD & AIM) Equine identification Ovine & caprine identification system Porcine identification |
|
| Area Management Team North and South |
Management, oversight and delivery of official controls In coordination with RVOs |
|
|
Eradication and Veterinary Medicines |
Eradication programmes (TB) Maintenance of official freedom of bovine brucellosis leucosis and brucellosis in sheep Authorisation, distribution and use of veterinary medicines |
|
|
Import Control Operations |
Management, oversight, and delivery of official controls in relation to import of live animals and products of animal origin at border control posts |
|
|
National Disease Control Centre |
Contingency planning and control of former OIE List A diseases Equine Policy Animal Health Surveillance |
|
|
Ruminant Animal Health |
Controls on trading premises (marts, dealers & assembly centres) |
|
|
Regional Veterinary Officers |
The Veterinary Service within the AMT carries out official controls |
|
|
Veterinary Export, Certification and International Trade |
Maintenance of export trade policy and veterinary export certification guidelines for EU and non-EU markets in livestock and livestock products Oversight and regulation of intracommunity pet and horse movement. Policy and oversight to ensure standards of animal welfare at transport are met Oversight, training, administration and support for DAFM use of TRACES Engagement at EU level in Potsdam meetings. |
Live Exports |
| Veterinary Medicines, AMR, ABP and TSEs |
VPHOps staff carry out the following activities in relation to TSE controls:
Regional veterinary office staff carry out the following activities in relation to TSE controls:
Staff from the NRL for TSEs carry out approvals and monitor the activities of rapid testing laboratories. Staff from the NRL for feed controls approve and monitor the activities of laboratories engaged in testing feed samples for meat and bone meal (MBM). Staff from Animal Feedingstuffs Division monitor the implementation of official controls relating to feed at import points, feed mills and on farm. Animal by-product official controls Other official activities relating to antimicrobial resistance Authorisation, distribution, and use of veterinary medicines Monitoring of residues of medicines and contaminants |
Veterinary Medicines and Medicated Feed |
|
Veterinary Public Health Operations |
Link to be added ---> Food of Animal Origin MANCP Page |
|
|
Veterinary Public Health Pig and Poultry Health |
Animal breeding controls Poultry trade Zoonoses |
Official Controls
Risk categorisation and frequency of official controls
The Head of Division (HoD) provides guidelines to field staff regarding the selection of premises for official control activity. Such premises are selected based on a combination of risk-based criteria and random assessment. In some cases, the premises to be subjected to official control are selected centrally based on certain risk-based selection criteria. Where possible, controls are carried out without prior warning. However, this may not always be possible, particularly if the business operator’s presence is required at the inspection.
A level of risk assessment is applied to the selection of premises for official control activity. The factors taken into account include the following:
- The nature of the business operator’s activity
- The volume of activity
- The interval since commencement of activity
- Results of previous inspections (if any)
- Compliance record of the operator
- Legislative considerations requiring specific levels of control
- Other local knowledge
Methods of official control
DAFM employs many different control methods and techniques while carrying out official controls. The methods employed are chosen based on their effectiveness and appropriateness, in consideration of the specific official control under examination. Often, multiple different techniques are used during official controls. They include inspection, surveillance, monitoring, interviews, document examination, verification of operator’s measurements, sampling, analysis, audits of operators
Risk based selection are based on the disease risk of an establishment, combined with the premise’s previous history of non-compliances. Some inspections are set at specific times intervals.
Recording of official controls
Animal Health and Import Policy Division (AHIPD) records performance and outcomes of official controls in TRACES. These results may also be accessed in TRACES.
Written records of official controls are recorded using the AFIT (Agriculture Field Inspection Testing) computer system.
National control plans
|
Division |
Official Control Area |
Specific control Programmes |
|
Animal Health and Import Policy |
International Trade |
|
|
Animal Identification and Movement |
Bovine ID |
|
|
Eradication |
Maintenance of Official Freedom of Bovine brucellosis, leucosis and brucellosis in sheep |
|
|
Eradication programmes (TB & Br) |
|
|
|
National Disease Control Centre |
Contingency planning and control of former OIE list A diseases |
|
|
Ruminant Animal Health |
Trading premises |
|
|
Veterinary Export, Certification and International Trade |
International Trade |
|
|
Veterinary Medicines, AMR, ABP and TSEs
|
TSEs and ABPs |
|
|
||
|
Monitoring of residues of medicines and contaminants |
|
|
|
Veterinary Public Health Pig and Poultry Health |
Animal Breeding (semen, ova and embryos) |
|
|
Trichinella |
|
|
|
Zoonoses |
|
National control plans and programmes are integrated into DAFM’s control systems in the following ways:
Each division performs the following tasks where appropriate:
- Arrange introduction of new legislation
- Authorisation of staff
- Provision of standard operating procedures, instruction manuals etc
- Provision of inspection forms (hard copy or electronic)
- Provision/organisation of training
- Co-ordination of activities with other relevant bodies
- Checks/systems to ensure the effectiveness of the control system
- Provide direction regarding official control activity for a given period
- Prescribe enforcement measures and sanctions
Resources and Facilities
Staffing resources
The divisions under the responsibility of the Chief Veterinary Officer had 1197 staff (1174.9 FTE) in August 2022. These staff work across 15 divisions, listed below, at several different grades in the administration, technical, veterinary and inspectorate streams.
One Health One Welfare Divisions under the responsibility of the Chief Veterinary Officer:
- Animal Health & Import Policy
- Animal Identification & Movement
- Animal Welfare Division
- Business System Co-Ordination & Development
- ERAD/Veterinary Medicines
- Import Controls Operations
- Milk & Meat Hygiene & Animal By-Products
- National disease Control Centre
- Ruminant Animal Health
- Vet Export, Certification & International Trade
- Vet Med, AMR, ABPs & TSEs
- Veterinary Public Health, Pig and Poultry
- Veterinary Public Health Operations
- Area Management Team North
- Area Management Team South
Each division assess their training needs at the start of each calendar year. Training needs are decided by (i) assessing the gap that exists in a division between the skills that exist and those that are required to fulfil their business needs during the year (ii) Team requests (iii) Individual staff member requests or training needs identified through their annual “Performance Management and Development System” return.
A Learning Needs Analysis (LNA) form is submitted by each division to the learning and development unit as an official record, stating the training required, which team members require it and the timeframe in which the division requires it to be delivered.
Laboratory resources
DAFM Laboratories provide laboratory support (bacteriology/parasitology, pathology and virology) for national disease eradication program
|
DAFM Laboratories Backweston |
Backweston, Celbridge, Co Kildare. |
|
Irish Equine Centre |
Testing of biological samples for certain notifiable diseases e.g., Equine Infectious Anaemia |
IT resources
All officers carrying out official controls have access to appropriate IT hardware, as required (computer, printers, laptops, mobile phones, tablets, videoconference facilities) , with effective broadband connections. Various databases and computer programs are used to record official controls. These include:
- AIM (Animal Identification and Movement)
- AFIT (Agriculture Field and Inspection testing)
- AHCS (Animal Health Computer System)
Officers also have access to TRACES as required.
DAFM also hosts an intranet, (Ezone) where up-to-date documented procedures can be published, contact details for colleagues can be found and this also hosts the DAFM organogram. This available for all officers.
IT resources are supported by a dedicated IMT division who develop, implement and maintain management information systems, and provide technical support to business areas.
Other resources
Other resources such as Personal Protective Equipment are provided as required to all DAFM officers.
Office facilities
Office facilities are managed in DAFM by a dedicated Accommodation division who provide suitable accommodation, utilities (non-IT), and supplies (non-IT). Office facilities are provided at central locations (such as Agriculture House and Backweston campus), regional offices (Regional Veterinary Offices) as well as offsite offices such as Dublin Port. They also ensure ISO 27001 certification.
Regulation (EU) 2017/625 (General Obligations)
Article 5(1)(a) Effectiveness and appropriateness of official controls
Staff within each division perform some checks to ensure that the official control system operates effectively. These checks may include:
- Review (both ongoing and end-of-year) of progress of the official control system. This can be obtained either directly from computer databases or, from data provided by the relevant DAFM managers. Feedback from middle managers regarding the outcome of official control activity is also taken into account
- On the spot visits by staff from DAFM to business operator premises
- Supervisory inspections by DAFM staff
- Follow-up of stakeholder feedback where relevant
- Outcome of re-performance inspections by integrated controls division
- Results of audits
- Monitoring by area management teams
- Laboratory feedback on sample numbers, quality and turnaround times
Article 5(1)(b) Consistency in the delivery of official controls
Consistency in delivery of official controls is achieved through training, documented procedures and regular liaison within DAFM divisions.
Article 5(1)(c) Ensuring staff performing official controls are free from conflicts of interest
Staff holding designated positions are required to complete conflict of interest forms each year so that any potential conflict can be managed appropriately where it arises.
Article 5(1)(d) Access to adequate laboratory capacity
The Department provides a wide range of specialist testing, diagnostic and reference laboratories embedded within its structure, headed up by the Head of Laboratories who is a member of the Department’s Management Board.
Article 5(1)(e) Access to suitably qualified and experienced staff
DAFM undertakes ongoing Workforce Planning & Succession Planning to provide clarity regarding processes for managing staff resources while facilitating continued organisational growth and positive performance across the Department. Recruitment of suitable staff is undertaken through a mix of competitions run by the Department and by the Public Appointments Service. Accurate job specifications and job descriptions are used in recruiting for the wide range of specialist positions within the organisation. All competitions feature appropriately experienced interviewers. Once in the organisation, new recruits are trained, supervised and subjected to probationary processes. Ongoing training and development help to ensure that staff can progress through the organisation.
Effective workforce planning and succession planning assists to ensure that a highly skilled workforce is maintained that are resilient, adaptable and equipped to deal with challenges in an ever-evolving work environment. Knowledge transfer is also key to planning for the future of the organisation and ensuring that the services we provide to internal and external customers continue to be delivered to the highest standards. Equally, encouraging the pursuit of learning and development opportunities amongst our staff is central to the maintenance of quality standards, morale and motivation.
Article 5(1)(f) Access to adequate facilities and equipment
The Information Systems Division of the DAFM is responsible for the maintenance and security of databases used by all divisions under the One Health, One Welfare business area. This division is also responsible for the provision of laptops, desktops and other IT equipment such as printers and scanners across all divisions in DAFM.
Article 5(1)(g) Ensuring staff have adequate legal powers to perform official controls
Veterinary officers are appointed as authorised officers under the Animal Health and Welfare Act 2013.
The activity within each division can be supported by several entities as follows:
- Legal Services Division (transposition, interpretation of legal issues, assessment of potential court proceedings etc)
- Investigations Division (investigation of legislative breaches sometimes with the view to prosecution)
- Centre for Veterinary Epidemiology and Risk Analysis (CVERA) (impact of disease etc)
Article 5(1)(h) Ensuring food business operators cooperate with staff performing official controls
Operators are legally obliged to assist an authorised officer in their duties as requested and the authorised officer has powers as laid down in S.I 187 of 2014 if the operator does not cooperate sufficiently.
Relationships, Memoranda of Understanding and Service Level Agreements with other Competent Authorities
The following Memoranda of Understanding (MoU’s) are in place:
|
Division |
Service Level Agreements or MOUs |
|
Animal Health and Import Policy |
Annual Service Contract (MOU) with FSAI Data Processing Agreement with ICBF MOU with AHI |
|
National Disease Control Centre |
MOU with DAERA in relation to cooperation between DAFM & DAERA where humane slaughter for the purpose of disease control (or animal welfare) is required |
|
Marine Institute |
Service Level Agreement (SLA) between DAFM and the Marine Institute (DAFM to carry out inspections of aquaculture operations) |
Communication procedures
Internal reporting and communication channels
Veterinary reporting arrangements are outlined in Figure XX below. The Agriculture Field Inspection & Test (AFIT) system is used increasingly for the scheduling, performance and reporting of official controls. Hard copy documents are used in areas where the AFIT system has not yet been introduced. Electronically captured data are readily available to management while data captured in hard copy format are reported to line management manually.
Where it is a requirement, specific (sector) reports are submitted to European Commission annually. DAFM contributes to the compilation of the MANCP annual report which summarises the control activity of the previous calendar year. The relevant data are forwarded to the FSAI for inclusion in the final report.
AHIPD records performance and outcomes of official controls in TRACES. These results may also be accessed in TRACES.
External reporting and communication channels
Formal:
- The Food Safety Liaison Unit (FLSU) provides for a formal communication system between DAFM and the FSAI
- DAFM divisions participate in relevant EU Commission working groups and committees as needed.
Import Policy:
- DAFM/DAERA Import Controls Liaison meetings
- EU Commission – DG SANTE (GREX, Audit, Communications)
- FSAI Service Contract Liaison meetings (annual)
- Other FSAI meeting as required, e.g., CCP meetings
- Revenue Customs
Cooperation between DAFM and the MI in relation to aquatic animal health is the subject of a formal service level agreement between DAFM and the MI. Formal meetings between the MI team and the relevant staff from DAFM are held at least twice yearly (typically one meeting in the first quarter to plan the years’ work and one meeting in the final quarter to review the years’ work).
On-going frequent liaison at management level in relation to work/policy issues take place between the Manager of the Fish Health Unit in the MI and the SVI/SSVI with responsibility for fish health issues within DAFM as required.
DAFM established a DAFM/LAVS Liaison Committee in July 2012. The objectives of the meetings are to have a forum for dealing with the wider aspects of Regulation (EU) 2017/625, including animal welfare and animal health. Discussions include:
- The Animal Health and Welfare Act 2013,
- Liaison with Regional Offices of DAFM on animal health and animal welfare - Animal welfare reporting,
- Control of Horses, animal health and welfare and control aspects,
- Updates on the discussion on the revision on Regulation (EU) 2017/625,
- Implementation of Regulation (EC) No 1099/2009 and training of LAVS on welfare matters under Regulation (EC) No 1099/2009,
- Animal welfare procedures for LAVS and,
- LAVS attendance at Better Training for Safer Food animal welfare training.
Documented Procedures
Availability of documented procedures to DAFM Staff
All official control activity is described in a standard operating procedure (SOP) or procedures manual which is prepared by the division with responsibility for the particular activity. Such procedures are regularly updated. Where necessary, relevant inspection forms are also prepared by the division and provided to field staff. Relevant notices and SOPs are available to staff on the DAFM intranet (Ezone).
Relevant documents when updated are placed on the intranet for all inspectors and staff to see and are followed up with a notice to staff alerting them to the updates. Updated documents are also shared with other agencies through the Cross Agency meetings described above as required.
Availability of documented procedures to the FSAI & Auditors
Documented procedures for official controls are supplied on request to organisations involved in auditing, usually as a result of pre-audit questionnaire process.
Reviewing & updating procedure
Documented procedures are reviewed and updated following updated/new legislation being published and as required following feedback from operators, inspectors and using any other relevant information received.
Audit
Internal audit
The Veterinary Internal Audit Unit (VIAU) performs audits in the One Health, One Welfare business line that comes under the responsibility of the CVO. The legal basis of the VIAU is Article 6 of Regulation (EU) 2017/625 and the VIAU follows the details laid out in Commission Notice on a guidance document on the implementation of the provisions for the conduct of audits under Article 6 of Regulation (EU) 2017/625 of the European Parliament and of the Council (2021/C 66/02). The VIAU’s primary role is auditing official controls and other official activities that fall within the scope of Regulation (EU) 2017/625. Oversight of VIAU activities is provided by the Audit Monitoring Group (AMG). The AMG is appointed by DAFM in accordance with the requirement of Article 6 of Regulation (EU) 2017/625 that audits shall be subject to independent scrutiny.
The VIAU produces a five-year audit programme which covers One Health, One Welfare areas in Regulation (EU) 2017/625. An annual programme is also produced annually. Audits are planned and prioritised using a risk-based methodology and taking results and plans for external audits into consideration.
External audit
DAFM is subject to external audits of official controls by DG Santé in accordance with Article 116 of Regulation (EU) 2017/625.
DAFM is subject to regular 3rd country audits of official controls in accordance with trading agreements and conditions of negotiated and agreed export certificates.
Contingency Planning and Mutual Assistance
Specific plans are in place to deal with incidents related to zoonotic disease, e.g., salmonella. Specific contingency plans are in place for List A diseases such as Foot and Mouth Disease (FMD), Newcastle Disease, Classical Swine fever, Avian Influenza and Bluetongue.
Documented procedures are in place for the management of on-farm animal welfare incidents, emergency slaughter and for large-scale slaughter on farm in the event of a disease outbreak
The National Disease Control Centre of DAFM has national contingency plans in place for the control and eradication of key exotic animal diseases including for example, Foot and Mouth disease, Avian influenza, African swine fever, Bluetongue, Rabies and others. As part of this contingency planning there are also specific operating procedures in place for the depopulation of animals for disease control purposes to be carried out in accordance with EU Council Regulation 1099/2009 on the protection of animals at the time of killing and Commission Delegated Regulation 2020/687. Such procedures are freely accessible to staff involved in outbreak control. Training of staff for the depopulation of animals for disease control purposes is provided by the National Disease Control Centre. In addition, DAFM has a Memorandum of Understanding with the Department of Agriculture, Environment and Rural Affairs in Northern Ireland for the provision of a specialised depopulation team to assist where necessary.
DAFM may communicate directly with a Member State (MS) in relation to a List B disease, e.g. Bovine TB. Contingency plans are also in place in respect of BSE in cattle and BSE in sheep. Divisions within DAFM have contributed to a business continuity plan outlining how critical activities may be carried out following the occurrence of unforeseen obstacles. As a minimum, in the case of an emergency in an area involving animal health or animal welfare, the Management Advisory Committee (MAC) of DAFM would coordinate the response. Contact points in the context of Regulation 1/2005 EC are used to communicate with other Member States.
Guidelines are in place in relation to managing acutely injured animals and during on-farm slaughter.
Problems regarding consignments of animals or animal products are addressed with the relevant member state as the need arises.
DAFM supports the concept of mutual assistance and is ready to provide such assistance as the need arises in line with the criteria contained in Directive 89/608 EC. Feed and food safety alerts are communicated to DAFM through the FSAI (RASFF). Animal health and welfare alerts are communicated through the CVO.
Ireland has agreed plans to mutually share staff with other countries in the case of animal health emergencies (USA, Canada, NZ, UK, Australia).
For further information on any matter raised on this page, or to suggest a correction, contact mancp@fsai.ie